Ionization Methods in Modern Mass Spectrometry
Samatha, Editorial team, Pharma Focus Europe
The analytical tool known as mass spectrometry (MS) serves as a potent method to detect chemical substance composition. MS instruments require ionization methods to convert molecular samples into charge-bearing compounds which allows for their subsequent analysis. This document evaluates different ionization methods employed in mass spectrometry by studying their structural concepts and practical applications as well as their benefits to understand their contemporary analytical chemistry importance.
Mass spectrometry (MS) stands as a fundamental analytical technique across multiple scientific disciplines because it serves vital functions in biochemistry, pharmacology, environmental science and also forensics. Through this method scientists achieve three primary outcomes which include substance identification together with molecular structure analysis and quantitative measurement of mixture components.
The MS process depends on ionization as its fundamental operational step. Different ionization methods need selection because the sample composition together with analytical sensitivity and data requirements shape which method to use. Mutual variations exist among ionization approaches regarding their fundamental principles and measurement efficiency and range of utilization. Scientists need to grasp different ionization methods because they determine which technique suits best for each mass spectrometry experiment. The article discusses principal ionization strategies for mass spectrometry alongside their operational foundations and utilization examples as well as evaluating their key benefits and constraints.

Ionization Techniques:
1. Electron Ionization (EI)
Electron Ionization (EI) represents the basic and popular ionization approach in mass spectrometry since it was first introduced during the 1950s. The technique entered mass spectrometry useages in the 1950s and continues to be essential because it provides highly reliable results and consistent outcomes.
The electron ionization process functions by using a powerful electron beam to strike molecules which exist in the gas phase. The molecular electron collisions lead to the ejection of an electron that creates a positively charged ion (M+). The mass spectrometer analyzes resulting molecular ions after they undergo fragmenting into smaller components.
This method proves most effective when dealing with small volatile compounds featuring hydrocarbons together with alcohols and aromatic compounds. Researchers deploy EI to analyze environmental samples as well as to test food items and beverages and to solve forensic cases.
Advantages:
Through EI we can obtain fragmentation patterns that automatically indicate unknown chemical structures.
The technique demonstrates historical reliability and possesses a stable foundation because it includes multiple spectra libraries used for analysis purposes.
Disadvantages:
EI fails to analyze big compounds and thermally sensitive or non-volatile chemical substances.
Analysis of the molecular ion through fragmentation usually results in profound dissociation that hinders exact weight determination of the analytical sample.

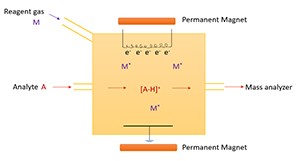
2. Chemical Ionization (CI)
The ionization technique referred to as Chemical Ionization generates results more gently than Electron Impact does. The technique provides ion generation for analysis with limited breakup of the analytical compound.
During CI process the introduction of methane or isobutane or ammonia serves as the reagent gas in the ionization chamber. The application of an electron beam within the reagent gas produces ionized molecules before they interact with the analyte through positive ions. During the reaction process analytes either accept or release a proton from their structure to become positively or negatively charged ions.
The operating range of CI includes analysis of sturdy complex molecules that EI cannot process effectively. The pharmaceutical field uses CI as their main method alongside petrochemical analysis for determining drug substances and metabolites in their research.
Advantages:
CI yields fewer analytical fragments than EI generates so analysis becomes more effective.
Larger molecules along with temperature-sensitive molecules that would experience excessive fragmentation in EI conditions can be analyzed through CI.
Disadvantages:
CI operates with higher selectivity than EI so it could fail to analyze specific compound types.
Nonetheless the ionization efficiency shows variations based on both the reagent gas type and the analyzed compound thus it provides less consistent outcomes when compared with EI.
3. Matrix-Assisted Laser Desorption/Ionization (MALDI)
Malasit is a "soft" ionization process which serves as a widespread method for biomolecular analysis of proteins and peptides along with nucleic acids.
During MALDI procedures the sample solution receives addition of a matrix compound that absorbs UV light as its primary constituent. When the mixture receives laser pulses the pulsed energy makes the matrix vaporize and ionize the sample. The laser energy helps produce sample molecule release into the gaseous state while ionization occurs.
The analytic technique MALDI serves numerous bioanalytical fields together with proteomics and genomics. The technique proves exceptionally useful for biomolecules such as proteins and oligonucleotides because it cannot detect these substances properly withEI or CI methods.
Advantages:
The MALDI technique generates little fragmentation during large biomolecule analysis thus enabling direct study of unbroken molecules.
The methodology offers efficient high-throughput operation because it effectively analyzes numerous samples present in blended mixtures at high speeds.
Disadvantages:
The analysis procedure of MALDI often depends on adding matrix compounds to specimens yet these compounds have potential detrimental effects on spectral interpretation.
The analytical sensitivity of this technique falls below other detection methods because of its limitations with trace molecule analysis.

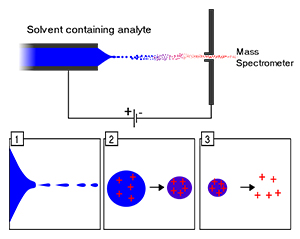
4. Electrospray Ionization (ESI)
ESI stands as an extensively applied ionization method for LC-MS analysis that specifically enhances investigation of big biomolecules together with polar compounds.
The ESI method requires researchers to dissolve their sample using solvent where they spray it using a thin needle under high voltage conditions. The procedure results in droplet formation followed by evaporation that generates charged ions during the transformation into gaseous particles. The produced ions undergo analysis using the mass spectrometer system.
Large biomolecules including proteins combined with peptides as well as nucleic acids and pharmaceutical compounds usually undergo analysis with ESI. The technique finds extensive use in environmental examination together with clinical applications.
Advantages:
The technological process of ESI works intrinsically soft and generates minimal molecular breaking so it preserves biomolecular structure cleanly.
The method establishes a connection with liquid chromatography to enable the study of complicated mixtures at both high sensitivity levels and resolution degrees.
Disadvantages:
ESI maintains limited compatibility with volatile compounds since it needs the sample to exist as a solution-based system.
Each analysis needs specific solvent and ionization optimization because these elements influence the performance of the technique.
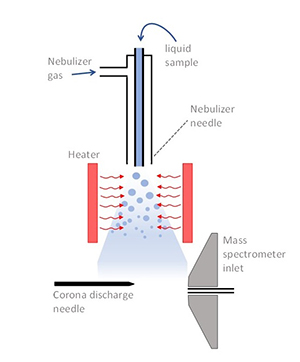
5. Atmospheric Pressure Chemical Ionization (APCI)
The technique of Atmospheric Pressure Chemical Ionization (APCI) takes the beneficial aspects of CI and ESI. APCI functions preferably for compounds with low polarities and low volatility which ESI does not optimize.
APCI uses an ionization chamber at atmospheric pressure which subjects the sample to a corona discharge to create ions from its analyte molecules. Mass spectrometry detects these generated charged species that arise from the sample introduction.
The analysis of pharmaceutical drugs and pesticide compounds together with environmentally unsafe pollutants demonstrates APCI's best application characteristics because of its capability to work with non-polar molecules between small and medium sizes.
Advantages:
The APCI method detects diverse compounds including polar along with non-polar substances through its analysis process.
This method provides decent detection capabilities because it works well with liquid chromatographic systems.
Disadvantages:
The analysis of proteins and highly polar compounds performs poorly when using the APCI method.
The ionization effectiveness of ions depends on the chemical composition of the examined molecule.
6. Ion Cyclotron Resonance (ICR)
The Ion Cyclotron Resonance (ICR) method operates as a high-resolution ionization method but needs Fourier transform mass spectrometry (FTMS) to deliver outstanding mass accuracy coupled with resolution.
ICR functions by locking ions within a magnetic field system that applies an electric field to let ions oscillate. Scientists measure oscillation frequencies in this technique to determine the mass-to-charge ratios of trapped ions based on the acquired frequencies.
ICR finds its main applications within high-resolution analytical workflows involving petrochemical tracing as well as molecular structure determination and measurements of isotopic ratios.
Advantages:
ICR achieves both high precision mass accuracy, together with outstanding resolution, so it defines unrivaled capabilities for intricate analytical purposes.
ICR-based spectroscopy supplies detailed spectra data that specialist researchers use to identify multiple substances in blended samples.
Disadvantages:
The implementation of ICR requires expensive specialized instrumentation, which also proves to be technically complex to operate.

Future Trends in Ionization Techniques:
The increasing needs for better sensitivity and faster analysis and higher specificity lead to the development of more advanced emerging ionization methods. DESI and DART represent key trends because they developed ambient ionization protocols which can conduct immediate field-based analyses without requiring extensive sample preparation steps. Scientists use these techniques more frequently to analyze forensic samples and for food testing and clinical medical diagnosis.
Nano-ionization methods are experiencing increased popularity particularly with nano-electrospray ionization (nano-ESI) because this technique delivers better ionization capabilities and decreased sample requirements that suit both proteomics and metabolomics applications. Soft ionization platforms are enhanced to minimize sample fragmentation while maintaining molecular structures throughout the analytical procedure.
The hybrid ionization approaches that join performance elements of present methods to create a solution for complex sample analysis by providing complete data. The combination of artificial intelligence and machine learning systems with ionization techniques through real-time parameter optimization will enhance reproducibility along with accuracy levels.
The future development of ionization methods will focus on improving their sensitivity together with portability along with automation capabilities to serve current scientific requirements and healthcare needs and environmental supervision objectives.
Conclusion:
Mass spectrometry ionization methods serve as fundamental tools to produce molecule ions which allows for identification and evaluation within the mass spectrometer. The different ionization procedures offer separate advantages and disadvantages which determine their suitability within particular analysis scenarios. The ability to choose the correct analytical technique depends on scientists gaining comprehension of various techniques and associated applications along with their strengths. Analytical technologies show promise for developing improved ionization methods and enhanced performance of mass spectrometry as scientists pursue better sensitivity accuracy and versatility across various scientific domains.


















