Accelerating the Drug Development Process
Mena Abdelsayed, Research Assistant Professor, Lankenau Institute for Medical Research
Kyriakos Kachrimanis, Department of Pharmaceutical Technology, School of Pharmacy, Aristotle University of Thessaloniki, 54124 Thessaloniki, Greece
Joerg Birkenfeld, Chief Scientific Officer, BioCopy AG
Synergy between high throughput-pHLA (peptide Human Leukozyte Antigen) screening technology on the one hand and AI-driven antibody design with end-to-end automated protein optimization accelerates the development of next-generation T-cell engagers for previously unexplored tumor targets.

The body's immune system exhibits a remarkable ability to scrutinize a cell's interior, a function of central importance. This scrutiny enables the detection of changes indicative of the transformation from a normal cell to a rapidly multiplying cancer cell. The human immune system has developed an intricate mechanism where every intracellular protein is presented on the cell's surface for immune surveillance. Proteins are broken down into peptides within the cell, binding to human leukocyte antigen (HLA) molecules and forming peptide HLA complexes (pHLA). If the amount of presented peptide is altered or unusual peptides are presented, cytotoxic T-cells recognize it through their T-cell receptors, selectively eliminating the transformed tumor cell.
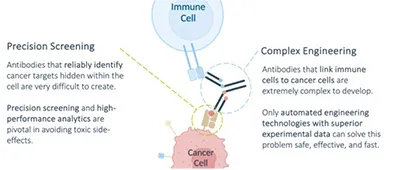
Despite being the most abundant surface markers on cancer cells, pHLA complexes present challenges for targeted cancer immunotherapies due to variable expression levels and marginal distinctions from those found in normal cells. This complicates the development of pHLA-targeting therapeutics, especially bi-specific T-cell engagers (TCEs) designed to redirect cytotoxic T-cells towards tumor sites. (Figure 1).
First, TCEs must precisely target cancer-specific pHLA complexes with the utmost specificity to avoid off-target effects, preventing severe toxicity in the patient. Second, the pHLA-targeting moiety needs to be combined with a T-cell targeting moiety to form a bi-specific modality. This is commonly achieved by engineering two distinct monoclonal antibodies — one directed at the cancer-specific pHLA complex and the other at a T-cell-specific protein like CD3 — into a single bi-specific antibody. Since bi-specific T-cell engaging antibodies do not naturally occur, their technical development requires extensive protein engineering efforts. The goal is to achieve a manufacturable, stable, and soluble compound which can be produced in substantial quantities for medical applications. The optimization of bi-specific T-cell engagers for pre-clinical and clinical development is a process that typically needs several years.
This combination of technologies may well be a transformative shift in the progression of next-generation pHLA-targeting T-cell engagers, aiming for superior safety and increased efficiency. This commitment is driven by the utilization of modern screening technology for ultra-high throughput pHLA and the automated protein engineering platform for unparalleled speed and efficiency in drug optimization. Both technologies are seamlessly integrated within a closed loop that incorporates artificial intelligence, enhancing the predictive capability for the design of pHLA-targeting TCEs with optimal developability and highest safety standards.
Precision Screening
Achieving precise discrimination of cancer cells without affecting healthy tissue is crucial for the development of T-cell engagers (TCEs). This requires accurate characterization of the pHLA-targeting antibody, considering its binding properties not only to the target cancer pHLA but also to other pHLAs resembling the target structure. Specialized technology is essential to meet these demands.
Precision screening is key for evaluating antibody binding to pHLA complexes, leveraging advanced microsystems technology and cutting-edge microarray chips. These technologies ensure exceptional accuracy in characterizing the binding properties of antibodies to pHLA complexes. The screening chips embed thousands of pHLA complexes, facilitating rapid and precise measurement of antibody binding to all relevant pHLA complexes in the body, enhanced by highly specialized bioinformatic workflows.
To initiate the process, in silico predictions are generated to identify pHLA complexes closely resembling the target pHLA. Those predicted pHLAs, together with rationally designed pHLA complexes, undergo precise measurement. The gathered results are augmented with additional experimental and omics data, allowing a precise bioinformatic prediction for the toxicity profile of a pHLA-targeting TCE. This risk assessment serves as a basis for informed decision making in the drug development process regarding safety and effectiveness.
Data-driven Drug Discovery Process
This technology serves as a cornerstone of developing next generation TCEs. The measurement data enables AI-driven predictions, establishing a closed loop that facilitates comprehensive antibody characterization and optimization across multiple cycles, all powered by the automated platform.
High-throughput Optimization of Bi-specific T-cell Engagers
In addition to recognizing the target pHLA molecule on cancer cells, the new antibodies must activate the body's immune cells against the identified tumor. To achieve this goal, the pHLA-binding antibodies are transformed into more complex structures known as bi-specific antibodies. Those antibodies possess a risk of unsuitability for development due to challenges in meeting industrial requirements for large-scale production, stable storage, and eventual use as a formulable drug. This often results in the failure of complex molecule developments at early stages.
These challenges can specifically be addressed with a fully automated and AI-powered platform for antibody optimization (2). The platform initiates the drug optimization process by in silico designing tens of thousands of bi-specific antibody variants. It seamlessly integrates the in-silico 'digital design' stage with the physical wet-lab stages within an end-to-end automated process. Using a parallelized approach, the automated platform produces up to 25,000 individual in silico-designed bi-specific antibody molecules. Each molecule is expressed in its desired soluble drug-product format within an isolated mini-bioreactor-like compartment, allowing simultaneous unbiased assessment of multiple functional and biophysical properties crucial for technical development, including yield, potency, stability, and solubility for each molecular design.
The automated platform accelerates drug development by more than 24 months, providing a substantial advantage over traditional optimization approaches. The platform's unique ability to preserve data on all variants ensures an accelerated learning, invaluable for refining the drug development process. As the process mirrors the final industrial drug production process, identified candidates undergo de-risking for progression into large-scale production and subsequent development through commercial manufacturing. The foundational concepts and technologies have been developed over many years by leading scientists, validated within the Big Pharma environment.
In order to ensure that the optimized candidates meet safety requirements, the physical data from the optimization platform are fully integrated with the off-target pHLA screening data. This combination enhances the identification of developable bi-specific antibodies. Furthermore, it enables the deep understanding of design principles underlying those antibodies, leading to enhanced efficacy and improved developability while maintaining high molecular safety standards. These insights are systematically incorporated into subsequent optimization cycles, streamlining the overall development process. This approach accelerates the identification of developable drug candidates from years to months compared to standard semi-automated and iterative design approaches. Additionally, it generates data sets of unprecedented quality and quantity and enables training of artificial intelligence for predicting improved designs of bi-specific pHLA-targeting TCEs with enhanced safety and efficacy.
Platform

Data Authority for Training AI-Models
The combination of these two technologies generates a unique "data authority," providing the capability to develop proprietary artificial intelligence (AI).
This integration also enables the development of new AI-based methods to accurately predict the design of novel pHLA-targeting TCEs. Harnessing AI capabilities empowers drug makers to precisely predict off-target toxicity and the developability of complex bi-specific antibodies. As a result, development timelines can be reduced significantly, and development processes will be streamlined continuously.
Conclusion
In summary, the innovative approach to combine precision screening, high-throughput engineering, and AI integration, can lead to a paradigm-shift in next-generation biologics development, particularly in immuno-oncology. The technologies promise enhanced safety, efficiency, and reduced development timelines so that patient safety can truly be prioritized and drug development may even be revolutionized.
(1) Krämer, S. et al. Sci Rep. 2023 Mar 31;13(1):5290. doi: 10.1038/s41598-023-32384-z.
(2) Furtmann, N. et al. Mabs . 2021 Jan-Dec;13(1):1955433. doi: 10.1080/19420862.2021.1955433.
(3) roPROTix (Biopharma Dealmakers (Biopharm Deal) ISSN 2730-6283
















