The Rise of Bioprinting: Opportunities and Challenges for Personalized Sterile Dosage Forms
Mostafa Essam Eissa, Independent Researcher and Consultant, Bioinformatics and Biometry Department, Pharmaceutical Research Facility, Cairo
Personalized medicine demands tailored treatments, but traditional drug manufacturing struggles to keep up. Bioprinting emerges as a potential solution, offering precision needed for customized dosage forms. We examine potential benefits and obstacles of using bioprinting for personalized medication.
Introduction
Personalized medicine, also known as precision medicine, has gained significant traction in recent years, transforming the healthcare landscape. By considering an individual's unique genetic, molecular, and environmental factors, personalized medicine aims to provide targeted therapies that maximize treatment effectiveness while minimizing adverse effects. This approach has shown remarkable success in various therapeutic areas, including oncology, cardiovascular diseases and infectious diseases.
Despite the advancements in personalized medicine, the manufacturing of sterile dosage forms, such as tablets, capsules, and injectables, has not kept pace. Traditional manufacturing methods often rely on mass production techniques that lack the flexibility and precision required to create personalized dosage forms with tailored drug combinations, release profiles, and dosage strengths. These limitations can hinder the full potential of personalized medicine, as patients may not have access to the most effective and individualized treatment options.
Bioprinting: A Promising Solution
Bioprinting, an additive manufacturing technique that utilizes living cells and biomaterials to create three-dimensional (3D) structures, has emerged as a promising solution for the development of personalized sterile dosage forms. By precisely depositing cells and biomaterials in a layer-by-layer fashion, bioprinting can fabricate customized dosage forms with intricate designs and controlled drug release profiles.
Opportunities of Bioprinting for Personalized Sterile Dosage Forms
Bioprinting offers several compelling opportunities for the advancement of personalized sterile dosage forms (Table 1):
1. Tailored Drug Combinations: Bioprinting enables the incorporation of multiple drugs into a single dosage form, allowing for personalized drug combinations based on an individual's specific needs. This approach can improve treatment efficacy and reduce the risk of drug interactions.
Table 1: Advantages of Tailored Drug Combinations using Bioprinting.
| Feature | Advantage | Example |
| Improved Efficacy | By combining drugs with synergistic or complementary mechanisms of action, bioprinting can create more effective treatments than single-drug therapies. | A bioprinted dosage form combining an anti-cancer drug with an immune checkpoint inhibitor* can enhance tumor targeting and immune response. |
| Reduced Side Effects | Bioprinting allows for the precise co-localization of drugs within a dosage form, potentially reducing systemic exposure and minimizing side effects associated with individual drugs. | A bioprinted dosage form containing an antibiotic and a probiotic can deliver targeted treatment to the gut while mitigating antibiotic-induced gut microbiome disruption. |
| Decreased Drug Interactions | By controlling the release profiles of multiple drugs from a single dosage form, bioprinting can minimize the risk of interactions that can occur when drugs are administered separately. | A bioprinted dosage form with a controlled-release antihypertensive drug followed by a vasodilator can ensure optimal blood pressure control without compromising blood flow. |
2. Controlled Release Profiles: Bioprinting can create dosage forms with controlled drug release profiles, ensuring that the medication is delivered at the optimal time and location within the body. This can enhance treatment effectiveness and minimize side effects. Figure 1 depicts the following:
Immediate Release: For rapid onset of action, bioprinted dosage forms can be designed to release the drug immediately upon administration. This is suitable for drugs that need to act quickly, such as pain relievers or allergy medications.
Sustained Release: Bioprinting can create dosage forms that release the drug over a prolonged period, improving patient compliance and reducing dosing frequency. This is beneficial for chronic conditions requiring long-term medication, such as hypertension, diabetes or asthma.
Pulsatile Release: By incorporating biomaterials with varying degradation rates or utilizing microfluidic channels within the bioprinted structure, pulsatile release profiles can be achieved. This approach mimics the natural release patterns of some hormones in the body, potentially leading to more effective and physiological treatment outcomes.
3. Patient-Specific Dosage Strengths: Bioprinting facilitates the fabrication of dosage forms with precise drug concentrations tailored to an individual's weight, metabolism, and drug sensitivity. This personalization can optimize treatment outcomes while reducing the risk of overdose or underdosing. Table 2 summarizes these patient-oriented benefits.
Table 2: Benefits of Patient-Specific Dosage Strengths achieved through Bioprinting.
| Feature | Benefit | Example |
| Improved Treatment Efficacy | By delivering the optimal drug dose based on individual factors, bioprinting can ensure that patients receive the most effective treatment possible. | A child with cancer can receive a bioprinted dosage form containing a chemotherapy drug at a safe and effective concentration adjusted for their weight and body size. |
| Reduced Risk of Adverse Effects | Personalized dosing through bioprinting can minimize the risk of overdosing, which can lead to severe side effects or toxicity. | A patient with a genetic variation affecting drug metabolism can receive a bioprinted dosage form with a lower concentration of a medication to avoid potential adverse reactions. |
| Enhanced Patient Compliance | Bioprinting can create dosage forms with personalized dosing schedules, potentially improving medication adherence, especially for complex treatment regimens. | An elderly patient with multiple chronic conditions can receive bioprinted dosage forms combining various medications at appropriate doses, simplifying their medication schedule. |
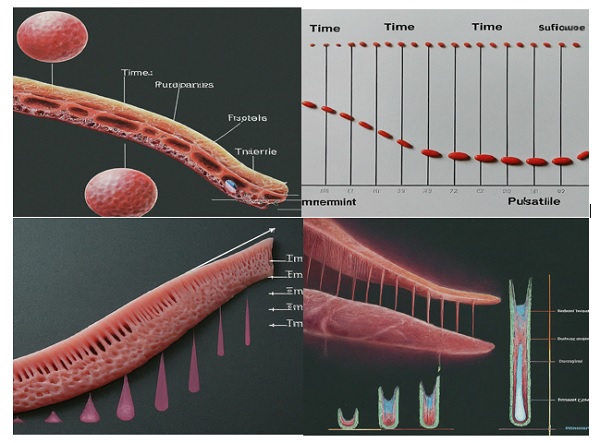
Figure 1: Schematic Illustration of Controlled Drug Release Profiles achievable with Bioprinting
4. Complex Geometries: Bioprinting allows for the creation of dosage forms with complex geometries, such as multi-layered structures or embedded drug depots, which can further control drug release and improve patient compliance. Figure 2 illustrates the following concepts:
- Multi-layered Structures: Bioprinting enables the creation of dosage forms with multiple layers, each containing different drugs or biomaterials. This allows for the controlled release of multiple drugs with distinct pharmacokinetic profiles from a single dosage form.
- Embedded Drug Depots: Bioprinting can fabricate dosage forms with embedded drug depots surrounded by biocompatible materials. These depots can release the drug over a sustained period or upon specific triggers, such as changes in pH or temperature.
- Bioprinted Implants: Bioprinting holds promise for the development of implantable dosage forms that can deliver drugs locally and continuously over extended periods. These implants can be designed with porous structures to facilitate cellular infiltration and potentially promote tissue regeneration alongside drug delivery.
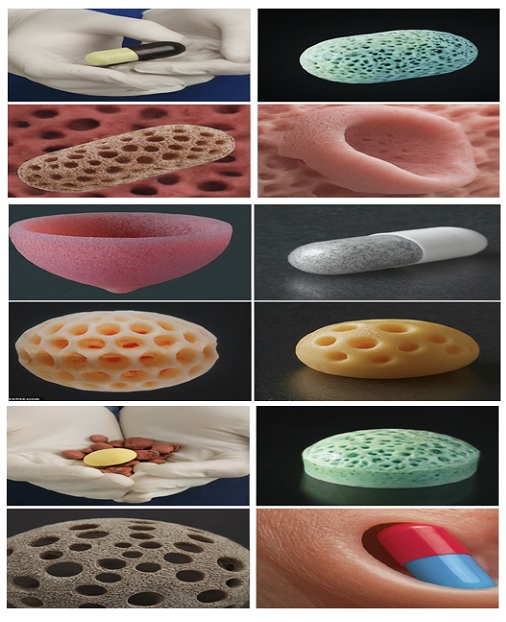
Figure 2: Examples of Complex Geometries achievable with Bioprinting for Personalized Sterile Dosage Forms
Challenges in Bioprinting Personalized Sterile Dosage Forms
Despite the promising opportunities, bioprinting for personalized sterile dosage forms faces several challenges:
- Scalability: Transitioning bioprinting from research setting to large-scale manufacturing requires significant advancements in automation, process control, and regulatory compliance. Current bioprinting processes are often slow and labor-intensive, limiting their applicability for high-throughput production of personalized medications. Strict quality control measures are essential to ensure the consistency and sterility of bioprinted dosage forms. Regulatory frameworks need to be adapted to address the unique aspects of bioprinted medicines, including safety assessments and approval processes for personalized therapies.
- Biomaterial Selection: Developing biocompatible, biodegradable, and printable biomaterials with the desired mechanical properties and drug release characteristics is crucial for successful bioprinting. Biomaterials used for bioprinting need to be compatible with living cells and not elicit adverse immune responses within the body. The biomaterial should ideally degrade naturally over time after delivering the therapeutic payload. The printability of the biomaterial is essential, ensuring it can be precisely deposited and retain its shape during the bioprinting process. Tailoring the biomaterial properties to achieve specific drug release profiles is an ongoing challenge requiring continued research and development.
- Sterilization Methods: Effective sterilization techniques are essential to ensure the sterility of bioprinted dosage forms without compromising the integrity of the bioprinted structures or the encapsulated drugs. Traditional sterilization methods, such as high heat or radiation, can damage living cells and biomaterials used in bioprinting. Novel sterilization techniques that are compatible with bioprinted constructs are being explored, such as ultraviolet (UV) irradiation or aseptic processing techniques.
- Regulatory Framework: Establishing clear regulatory guidelines and standards for bioprinted dosage forms is necessary to ensure patient safety and facilitate their approval and commercialization. Regulatory bodies need to develop frameworks that address the unique characteristics of bioprinted medicines, considering factors such as biomaterial safety, process control, and personalized dosing. Collaboration between researchers, manufacturers and regulatory agencies is crucial to establish clear pathways for clinical trials and approval processes for bioprinted medicines.
Future Directions
The field of bioprinting for personalized sterile dosage forms is rapidly evolving, with several exciting areas of future exploration:
- Integration with Microfluidics: Bioprinting can be integrated with microfluidic technologies to create complex microchannels within dosage forms. These microchannels can facilitate the delivery of multiple drugs with precise spatial and temporal control, further enhancing the capabilities of bioprinted medicines.
- Bioprinting with Stem Cells: Bioprinting holds promise for the development of regenerative therapies that combine drug delivery with stem cell implantation. By incorporating stem cells into bioprinted scaffolds, researchers aim to create functional tissues that can repair or replace damaged organs.
- Bioprinting for In Vitro Diagnostics: Bioprinting can be utilized to create customized tissue models for in vitro diagnostics. These bioprinted models can be used to study drug response in a patient-specific manner, aiding in personalized treatment selection and drug development.
Conclusion
Bioprinting holds immense promise for the development of personalized sterile dosage forms, revolutionizing the way we manufacture and deliver medications. By addressing the current challenges through continued research and development, bioprinting has the potential to transform personalized medicine, providing patients with access to truly individualized and optimized treatment options. The combined efforts of researchers, engineers, clinicians, and regulatory bodies will be essential to overcome the existing hurdles and usher in a new era of bioprinted medicines for improved patient care. In conclusion, bioprinting offers a revolutionary approach to personalized medicine by enabling the fabrication of sterile dosage forms tailored to individual patient needs. By overcoming the existing challenges and exploring promising future directions, bioprinting has the potential to significantly improve treatment outcomes and redefine the landscape of healthcare.













