mRNA and Oligonucleotide Therapeutics: Emerging Synergies in Genetic Medicine
Dr. Prasad Panzade, PhD, President & Chief Scientific Officer, Panztecs Inc.
Messenger RNA (mRNA) and synthetic oligonucleotide therapeutics have emerged as transformative platforms in genetic medicine, enabling modulation of gene expression, correction of pathogenic mutations, and targeted delivery of therapeutic payloads. While historically developed as separate modalities, recent advances in chemistry, delivery systems, and clinical translation are revealing strong synergies between the two fields. This review explores the complementary mechanisms, overlapping technological innovations, and combining therapeutic potential of mRNA and oligonucleotides, with emphasis on delivery strategies, disease targets, and future directions in precision medicine.
Genetic medicine has rapidly evolved with the advent of nucleic acid therapeutics that modulate gene expression beyond traditional protein and small molecule drugs (Burnett et al., 2018). Two leading modalities, messenger RNA (mRNA) and synthetic oligonucleotides offer distinct yet complementary approaches to treat a range of diseases.
mRNA therapeutics involve the delivery of engineered mRNA sequences into cells to enable the transient expression of therapeutic proteins, vaccines, or gene-editing tools. Meanwhile, oligonucleotide therapeutics, including antisense oligonucleotides (ASOs), small interfering RNAs (siRNAs), microRNA mimics/inhibitors, and aptamers, modulate endogenous RNA transcripts to silence or correct gene expression without altering the genome.
The recent success of mRNA COVID-19 vaccines alongside FDA approvals of oligonucleotide drugs such as nusinersen and inclisiran underscores the clinical viability and potential convergence of these platforms
Mechanistic Complementarity
mRNA Therapeutics
mRNA therapies work by introducing synthetic mRNA encoding a therapeutic protein, enabling transient protein expression that can replace deficient enzymes, stimulate immune responses, or facilitate gene editing. This additive approach is beneficial for diseases caused by loss-of-function mutations or when temporary protein expression is desired.
Oligonucleotide Therapeutics
Oligonucleotides regulate gene expression via sequence-specific binding to RNA, resulting in transcript degradation (via RNase H or RNA-induced silencing complex [RISC]) or modulation of splicing. This subtractive or modulatory approach is effective for dominant-negative mutations or toxic gain-of-function effects.
Synergy Opportunities
Combining these mechanisms opens possibilities for comprehensive gene regulation: mRNA can restore deficient protein function, while oligonucleotides can suppress pathological transcripts in the same disease pathway. Additionally, delivery advances developed for one modality can accelerate the other, such as lipid nanoparticle (LNP) systems or ligand conjugation strategies.
Shared technologies foundations
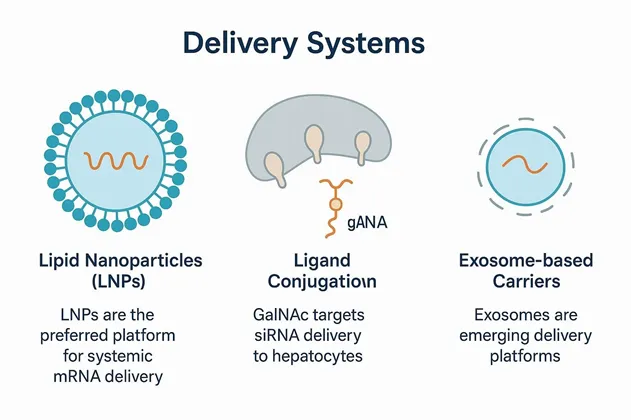
Delivery Systems
Lipid nanoparticles (LNPs) have become the preferred platform for systemic mRNA delivery, as exemplified by COVID-19 vaccines, and are being adapted for oligonucleotide delivery with optimised lipid composition and charge ratios.
Ligand conjugation approaches such as N-acetylgalactosamine (GalNAc) targeting for hepatocyte-specific siRNA delivery illustrate successful receptor-mediated uptake that could be expanded to mRNA therapeutics for liver diseases. Emerging delivery platforms, including exosome-based carriers, also show promise for both modalities.
Chemical Modifications
Chemical modifications improve RNA stability and reduce immunogenicity. mRNA often incorporates modified nucleosides like pseudouridine and 5-methylcytidine to evade innate immune sensing and enhance translation. Oligonucleotides utilise phosphorothioate backbones, 2’-O-methyl, and locked nucleic acid (LNA) modifications to increase resistance to nucleases and affinity for target RNA. Cross-learning between these chemistries is ongoing to optimise both modalities.
Therapeutic Areas of Overlap
Emerging Trends and Future Outlook
Regulatory Convergence
The success of mRNA vaccines has accelerated regulatory acceptance of RNA-based drugs, providing a framework likely to benefit oligonucleotide approvals and combination RNA therapies.
Manufacturing Synergies
Manufacturing platforms for in vitro transcription, purification, and LNP formulation are increasingly modular and scalable, enabling cost-effective production of both mRNA and oligonucleotides. Advances in continuous manufacturing and single-use systems will further streamline development.
Personalized and Combination Therapies
Precision medicine approaches integrating genomic profiling allow for rapid design of bespoke mRNA and oligonucleotide cocktails tailored to individual patients, particularly for rare diseases and oncology. Combination therapies harnessing complementary mechanisms hold promise for improved efficacy and reduced resistance.
Conclusion
mRNA and oligonucleotide therapeutics are at the forefront of genetic medicine, each with unique advantages. Their mechanistic complementarity, shared delivery and chemistry innovations, and expanding clinical applications suggest a future of integrated RNA therapies that address complex diseases more effectively than either platform alone. Continued research, manufacturing advances, and regulatory alignment will be critical to fully realise the potential of these emerging synergies
References
- Baden, L. R., et al. (2021). Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. New England Journal of Medicine, 384(5), 403–416. https://doi.org/10.1056/NEJMoa2035389
- Bennett, C. F., et al. (2022). RNA Therapeutics: A Potential Game Changer for Rare Genetic Diseases. Annual Review of Medicine, 73, 1–18. https://doi.org/10.1146/annurev- med-042120-023729
- Burnett, J. C., et al. (2018). RNA-Based Therapeutics: Current Progress and Future Prospects. Chemistry & Biology, 25(2), 185–195. https://doi.org/10.1016/j.chembiol.2017.10.002
- Crooke, S. T., et al. (2021). Antisense Drug Technology: Principles, Strategies, and Applications. Pharmacological Reviews, 73(2), 345–377. https://doi.org/10.1124/pr.120.019594
- Finkel, R. S., et al. (2017). Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. New England Journal of Medicine, 377(18), 1723–1732. https://doi.org/10.1056/NEJMoa1702752
- Food and Drug Administration (FDA). (2021). Guidance for Industry: COVID-19 Vaccines — Development and Licensure.
- Hou, X., et al. (2021). Lipid Nanoparticles for mRNA Delivery. Nature Reviews Materials, 6(12), 1078–1094. https://doi.org/10.1038/s41578-021-00358-0
- Kalluri, R., & LeBleu, V. S. (2020). The Biology, Function, and Biomedical Applications of Exosomes. Science, 367(6478), eaau6977. https://doi.org/10.1126/science.aau6977
- Karikó, K., et al. (2008). Incorporation of Pseudouridine into mRNA Yields Superior Nonimmunogenic Vector with Increased Translational Capacity and Biological Stability. Molecular Therapy, 16(11), 1833–1840. https://doi.org/10.1038/mt.2008.200
- Kole, R., et al. (2012). Antisense Oligonucleotides: From Design to Therapeutic Application. Nature Reviews Drug Discovery, 11(2), 125–140. https://doi.org/10.1038/nrd3625
- Li, C., et al. (2021). Antiviral siRNA Delivery Strategies to the Respiratory Tract. Advanced Drug Delivery Reviews, 179, 114001. https://doi.org/10.1016/j.addr.2021.114001
- Nair, J. K., et al. (2017). Multivalent N-Acetylgalactosamine-Conjugated siRNA Localizes in Hepatocytes and Elicits Robust RNAi-Mediated Gene Silencing. Journal of the American Chemical Society, 139(28), 9931–9937. https://doi.org/10.1021/jacs.7b03473
- Pardi, N., et al. (2018). mRNA Vaccines, A New Era in Vaccinology. Nature Reviews Drug Discovery, 17(4), 261–279. https://doi.org/10.1038/nrd.2017.243
- Patel, S., et al. (2022). Advances in Manufacturing of mRNA Therapeutics: Challenges and Opportunities. Pharmaceutical Research, 39, 1679–1692. https://doi.org/10.1007/s11095-022-03255-0
- Polack, F. P., et al. (2020). Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. New England Journal of Medicine, 383(27), 2603–2615. https://doi.org/10.1056/NEJMoa2034577
- Ramaswamy, S., et al. (2022). Personalized RNA Therapeutics: Tailoring Nucleic Acid Drugs to the Individual Patient. Trends in Molecular Medicine, 28(3), 224–236. https://doi.org/10.1016/j.molmed.2021.11.004
- Rigo, F., et al. (2014). Antisense Oligonucleotides in Neurology. Nature Reviews Neurology, 10(7), 373–386. https://doi.org/10.1038/nrneurol.2014.81
- Sahin, U., et al. (2014). mRNA-Based Therapeutics , Developing a New Class of Drugs.
- Nature Reviews Drug Discovery, 13(10), 759–780. https://doi.org/10.1038/nrd4278
- Sahin, U., et al. (2017). Personalized RNA Mutanome Vaccines Mobilize Poly-Specific Therapeutic Immunity against Cancer. Nature, 547(7662), 222–226. https://doi.org/10.1038/nature23003
- Samaridou, E., et al. (2020). Lipid Nanoparticles for Nucleic Acid Delivery: Current Perspectives and Future Directions. Advanced Drug Delivery Reviews, 154-155, 37–63. https://doi.org/10.1016/j.addr.2020.06.011
- Wang, J., et al. (2021). Recent Advances in Oligonucleotide Therapeutics: Chemistry, Delivery, and Clinical Applications. Frontiers in Pharmacology, 12, 667516. https://doi.org/10.3389/fphar.2021.667516