EU Pharma Reform 2025 Explained: What Market Access Leaders Must Know for 2026-2029
The EU Pharma Reform is reshaping the pharmaceutical landscape across Europe. Following the Council of the EU's landmark March 2024 agreement, this article decodes the shift from fixed-term regulatory data protection to a performance-based system rewarding broad launch and unmet medical need (UMN) focus. It discusses how the reform will affect orphan drugs, antimicrobial resistance (AMR), and supply security and provides a strategic roadmap of market access, pricing, and launch planning in Europe starting in 2026.
Introduction: A Game-Changer for European Pharma
What if your EU market exclusivity depends on launching your medicine in every member state and proving its real-world value?
This is no longer hypothetical. The historic Pharma Reform agreement of the Council of the EU of March 2024 is the first major revision of pharmaceutical legislation in more than 20 years. It will have a significant impact on the way businesses will enter the market, set their prices, and launch strategies in Europe in 2026 and beyond.
These changes are not optional for pharma leaders and market access strategists—they're essential to future success
The Core Deal: Balancing Patient Access, Innovation & Competitiveness
The reform balances four main objectives:
- Improving patient access through the minimization of disparities among EU member states.
- Enhancing innovation, especially to address unmet medical demands, including antimicrobials and rare diseases.
- Improving supply security to prevent critical medicine shortages
- Supporting EU competitiveness to keep it attractive for pharma R&D
Although the final legislation will be negotiated with the European Parliament, the agreement of the Council will provide a clear way forward.
Key Changes & What They Mean for Market Access
1. New Incentives & Market Exclusivity Rules
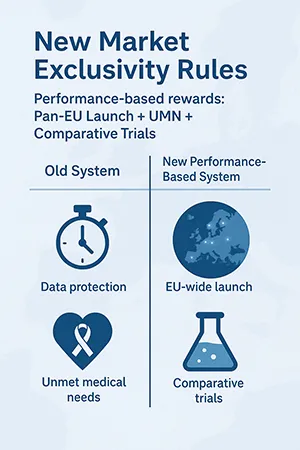
Performance based system substitutes the fixed 8+2+1 regulatory data protection interval.
| Protection Period | Old System | New System (Council) |
| Baseline Data Protection | 8 years | 6 years |
| Bonus for Launching in All EU | N/A | +2 years |
| Bonus for Addressing UMN | +1 year | +6 months |
| Bonus for Comparative Clinical Trials | N/A | +6 months |
| Bonus for New Therapeutic Indication | +6 months | +6 months |
Implications:
- The maximum exclusivity remains similar but is now conditional and variable.
- The +2 years for EU-wide launch incentivizes pan-European launches, ending the traditional staggered launch strategy focused on select markets.
- Demonstrating value through UMN and comparative trials is critical to unlock bonus protection, requiring market access to be involved early in R&D.
2. Orphan Drug Exclusivity Reform
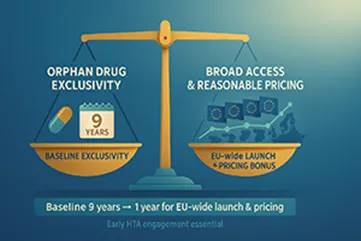
| Exclusivity Period | Old System | New System |
| Baseline | 10 years | 9 years |
| Bonus for EU-wide Launch & Pricing | N/A | +1 year (if reasonable & sustainable pricing) |
What This Means:
Orphan drugs must now link exclusivity incentives directly to broad access and pricing that reflects therapeutic value. High pricing without justification may cost a full year of market exclusivity. Engage HTA bodies and payers early.
3. Tackling Antimicrobial Resistance (AMR)
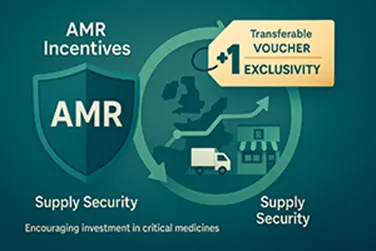
The reform introduces transferable data exclusivity vouchers:
- +1 year exclusivity voucher granted for new antimicrobials addressing critical unmet needs
- This voucher can be sold or used on another product, creating a financial incentive to invest in AMR research
Market Access Impact:
This new voucher system creates a novel revenue stream and shifts the competitive landscape, rewarding companies tackling one of Europe’s most urgent public health challenges.
4. Strengthening Supply & Shortage Preparedness
Key new obligations:
- Earlier shortage notifications
- Potential shortage prevention plans for critical medicines
- A public Union-wide list of shortages to improve transparency
Why It Matters:
Reliable supply chains are now an essential part of market access success. Noncompliance can result in penalties and damage payer relationships.
Strategic Action Plan for 2026-2029 Market Access Success

- Integrate Market Access Early in R&D
Design clinical trials that highlight unmet needs and comparative advantages to secure bonus exclusivity. - Plan Pan-European Launches
Build operational capacity to navigate multiple EU reimbursement systems simultaneously—staggered launches will cost exclusivity. - Master Value Demonstration & Storytelling
Create compelling value dossiers that justify pricing and clinical benefit to unlock incentives and satisfy HTA requirements. - Engage Early and Often with HTA Bodies & Payers
Understand evolving perspectives on reasonable and sustainable orphan drug pricing. - Monitor Trilogue Negotiations
Stay agile as the final legislation is shaped through ongoing EU Parliament discussions.
Conclusion: A New Era for EU Pharma
The EU Pharma Reform 2025 signals a move from fixed incentives to a dynamic, performance-based model. The market exclusivity rewards are still high but should be achieved by wide access, evident value creation, and launch superiority.
Current clinical trial design, launch planning, and payer engagement preparations will be the difference between your success in the new European pharmaceutical environment in 2026 and beyond.
Want to stay ahead on EU Pharma Reform updates?
Get the latest insights by subscribing to our newsletter or filling out the form.

















