Chiral Biosensors: Precision Tools for Safer Drugs
Dr. Selvakumar Kanthiah, Associate Professor, Acharya and BM Reddy College of Pharmacy
Dr. Valliappan Kannappan, Founder, Chiralpedia
In drug development, identifying the right mirror-image molecule - called an enantiomer - can be the difference between a life-saving treatment and unwanted side effects. Chiral biosensors are revolutionising this process by offering fast, accurate detection of these critical differences. By blending biology with smart sensing technologies, they’re helping researchers develop safer, more effective medicines. This article explores how these next-gen tools are advancing pharmaceutical quality and what’s on the horizon for this promising technology.
Introduction
The pharmaceutical industry has long been recognised the profound implications of chirality in drug design and development. Enantiomers, mirror-image isomers of a chiral molecule, show dramatically distinct pharmacokinetic (PK) and pharmacodynamic (PD) profiles. One may be therapeutically active, while its counterpart could be inactive or harmful. As a result, the precise detection, discrimination, and quantification of chiral compounds have become necessary in chiral drug development and regulatory compliance.
Traditionally, chiral separation and detection is based on chromatographic techniques, such as chiral HPLC or capillary electrophoresis. While effective, these methods are often resource-intensive and require extensive sample preparation, skilled analyst, and sophisticated instrumentation. The advent of chiral biosensors offers a transformative alternative, providing fast, cost-effective, and enantioselective detection with potential for real-time monitoring, on-site application. In this article, we delve into the basics and types of chiral biosensors, along with emerging technologies and materials driving their advancement. We highlight the growing demand and practical applications in pharmaceuticals. The discussion also covers current challenges and future directions shaping this innovative field.
Chiral biosensors, a new and innovative technology designed to distinguish between mirror-image molecules, paving the way for more efficient drug discovery, personalised medicine, and clinical diagnostics. A chiral biosensor is designed to detect and measure the presence of chiral compounds, through specific interactions. These sensors leverage the natural ability of biological recognition elements (such as enzymes, antibodies, or aptamers) to differentiate between enantiomers. The resulting detection can be in the form of electrochemical, optical, or mass-based signals that provide real-time, highly sensitive analysis of the enantiomeric composition of a sample.
It consists of three main components:
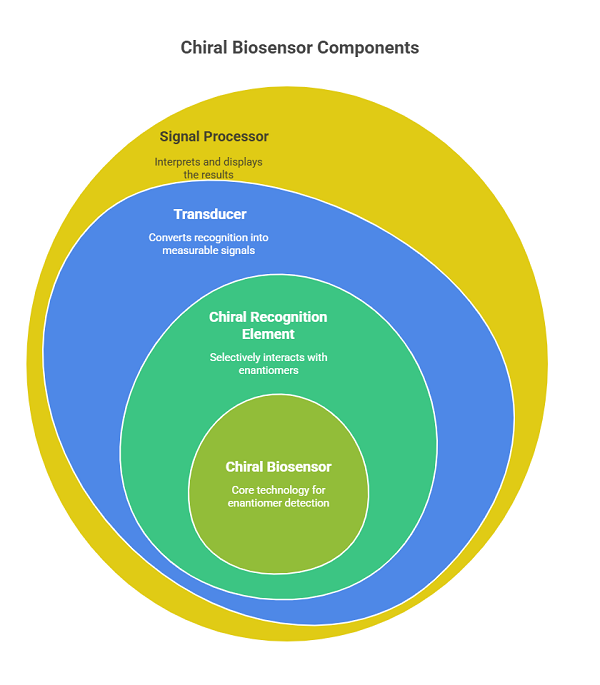
- Chiral recognition element: Selectively interacts with one enantiomer over the other (e.g., enzymes, antibodies, aptamers, chiral selectors).
- Transducer: Converts the recognition into a measurable signal (optical, electrochemical, piezoelectric, etc.).
- Signal processor: Interprets and displays the result.
The specificity of chiral biosensors arises from the 3D configuration of the recognition element, which mimics biological chiral discrimination observed in enzymes and receptors.
1. Types of chiral biosensors
 Chiral biosensors are classified by detection mechanisms and recognition elements, each offering unique advantages for pharmaceutical applications like drug screening, enantiomeric purity analysis, and clinical diagnostics. Key types include
Chiral biosensors are classified by detection mechanisms and recognition elements, each offering unique advantages for pharmaceutical applications like drug screening, enantiomeric purity analysis, and clinical diagnostics. Key types include
- Electrochemical sensors that detect electrical changes for quality control and drug monitoring.
- Optical sensors measuring circular dichroism, fluorescence, or SPR for high-throughput screening and real-time analysis.
- Mass-sensitive sensors like quartz crystal microbalance (QCM) for formulation and stability studies; aptamer-based sensors with high selectivity for personalised medicine.
- Enzyme-linked sensors using enantioselective enzymes for metabolism and toxicity studies.
- Molecularly imprinted polymer (MIP) sensors offering robust, cost-effective industrial QA/QC.
- Surface plasmon resonance (SPR) sensors adapted for real-time monitoring of chiral interactions, crucial in pharmaceutical R&D and bioscreening.
These technologies provide a versatile toolkit for advancing stereoselective detection across the pharmaceutical lifecycle.
2. Emerging technologies and materials in chiral biosensors
The integration of advanced materials and nanotechnology is driving a paradigm shift in the development of chiral biosensors, aligning with the pharmaceutical industry’s demand for high sensitivity, specificity, and portability in stereoselective analysis.
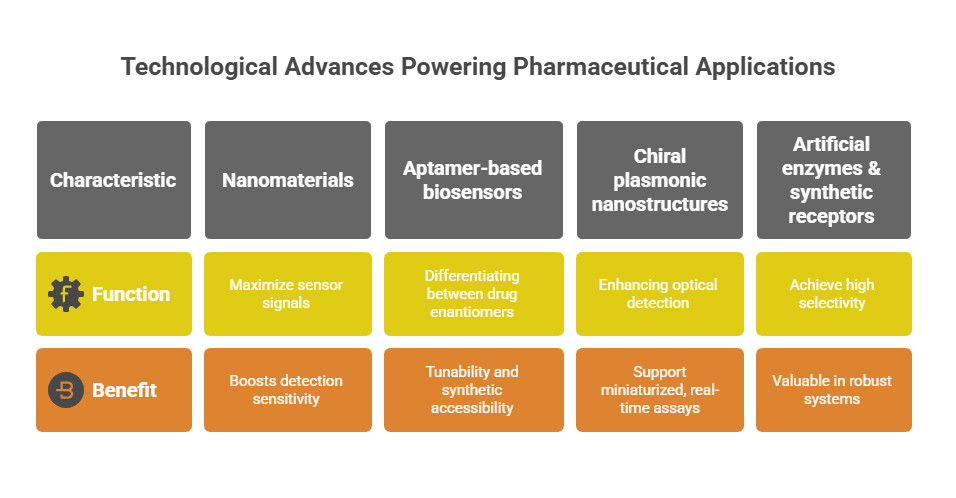 Key technological advances powering pharmaceutical applications:
Key technological advances powering pharmaceutical applications:
- Nanomaterials for enhanced sensitivity
Gold nanoparticles, graphene, and carbon nanotubes are being employed to maximize sensor signals and offer high surface-to-volume ratios. This enables efficient immobilization of chiral recognition elements and boosts detection sensitivity to femtomolar levels, essential for low-dose chiral drug monitoring and impurity profiling in final formulations.
- Aptamer-based chiral biosensors
DNA/RNA aptamers that adopt enantioselective 3-dimensional conformations are proving highly effective for differentiating between drug enantiomers. Their tunability and synthetic accessibility make them ideal for developing targeted biosensors for specific chiral APIs, especially in biopharmaceutical and peptide drug development.
- Chiral plasmonic nanostructures
Engineered plasmonic materials that interact differently with right- and left-handed enantiomers are enhancing optical detection in label-free formats. These platforms support miniaturised, real-time assays that could be integrated into manufacturing lines for in-process control of enantiomeric purity.
- Artificial enzymes & synthetic receptors
Biomimetic systems designed to replicate the enantioselectivity of natural enzymes are being incorporated into biosensors to achieve high selectivity without relying on delicate biological materials. These components are particularly valuable in robust, field-deployable chiral sensor systems suitable for remote quality assessment or decentralised trials.
By integrating these emerging technologies, chiral biosensors are becoming integral to pharmaceutical analytics, offering the precision and responsiveness needed in modern drug development and manufacturing pipelines.
3. The growing need for chiral biosensors
The significance of chirality in drug discovery cannot be overstated. The human body is inherently chiral, most biological systems, from enzymes to receptors, interact differently with each enantiomer of a drug. This means that, in many cases, only one enantiomer is therapeutically beneficial, while the other might be inactive or unsafe.
4.1 Practical applications
 The significance of stereoselective detection in the pharmaceutical industry is not merely theoretical, numerous marketed drugs exhibit enantiomer-specific therapeutic and adverse effects. For example, the thalidomide tragedy of the 1950s, where one enantiomer caused severe birth defects, highlights the need for careful enantiomeric screening during drug development. Chiral biosensors are now filling the gap left by traditional methods such as chromatography and spectroscopy, offering quicker, more efficient ways to analyze drug enantiomers. The applications of chiral biosensors in pharmaceutical spectrum includes
The significance of stereoselective detection in the pharmaceutical industry is not merely theoretical, numerous marketed drugs exhibit enantiomer-specific therapeutic and adverse effects. For example, the thalidomide tragedy of the 1950s, where one enantiomer caused severe birth defects, highlights the need for careful enantiomeric screening during drug development. Chiral biosensors are now filling the gap left by traditional methods such as chromatography and spectroscopy, offering quicker, more efficient ways to analyze drug enantiomers. The applications of chiral biosensors in pharmaceutical spectrum includes
a. Stereoselective screening in drug discovery
Chiral biosensors enable stereoselective screening of new drug candidates. For example, these biosensors can test which enantiomer of a drug binds more strongly to a target protein or receptor, helping researchers identify the most effective and safe candidate. This stereoselectivity is crucial for developing drugs that target specific receptors with high precision.
b. Enantiomer-specific drug delivery
Advanced chiral biosensor platforms are being used to design and evaluate drug delivery systems that release specific enantiomers at targeted sites, optimizing PKs and PDs while minimizing adverse effects.
c. Enantiomeric purity and quality control
One of the most important applications of chiral biosensors is in the quality control of drugs. Drug developers need to ensure that the active enantiomer of a drug is present in the correct concentration, while minimizing the inactive or harmful enantiomer. Chiral biosensors offer a real-time, efficient way to monitor enantiomeric purity during drug development and production, ensuring that only the therapeutic enantiomer is present in the final formulation.
d. Personalized medicine
Chiral biosensors allow for individualised treatment strategies by assessing how patients metabolise specific drug enantiomers. This enables clinicians to tailor dosing regimens that maximize efficacy and minimize toxicity, particularly for drugs with chiral-dependent metabolism.
e. Chirality-based drug target validation
Chiral biosensors are also used to validate stereospecific interactions between drugs and their selective biological targets. Understanding how a drug’s enantiomers interact with specific receptors or enzymes provides invaluable insight into its mechanism of action. This can help identify the optimal enantiomer for targeting a disease or condition.
f. Real-time process monitoring
Used in process analytical technology (PAT) frameworks for continuous monitoring of enantiomeric composition during synthesis and formulation.
4.2 Chiral Biosensors: Application Across the Pharmaceutical Value Chain
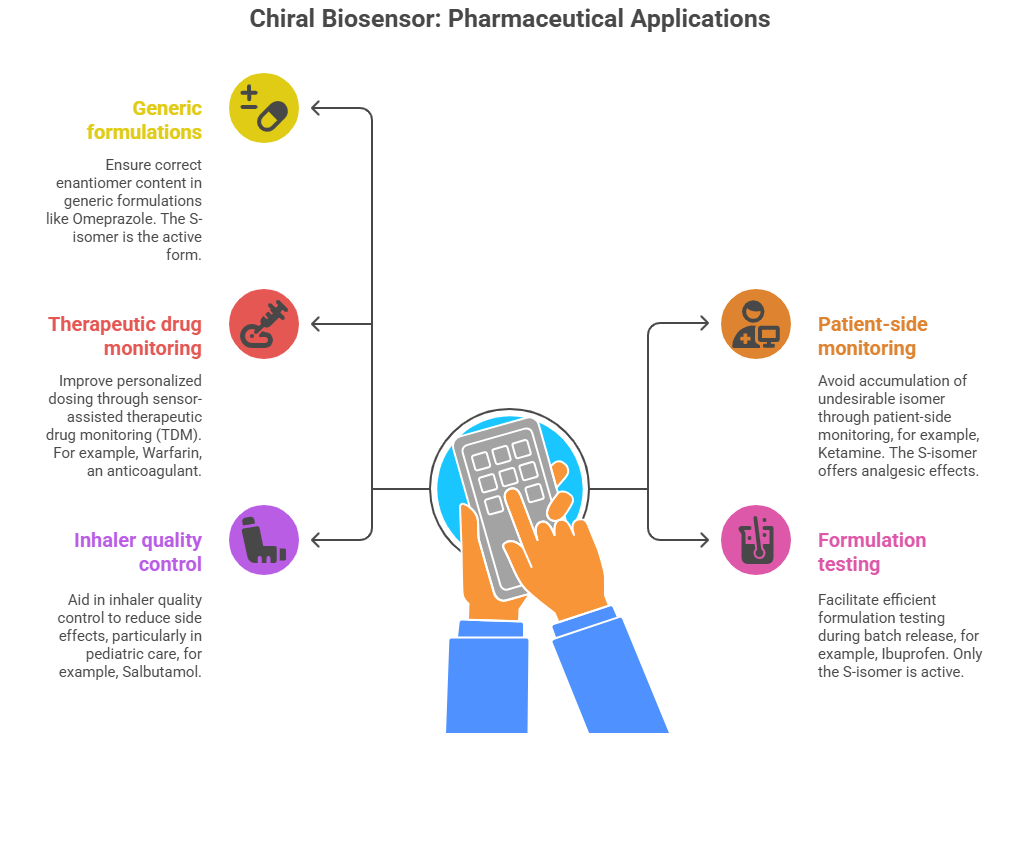 Chiral biosensors have significant implications across various drug classes by enabling precise enantiomer detection and monitoring.
Chiral biosensors have significant implications across various drug classes by enabling precise enantiomer detection and monitoring.
- Help ensure the correct enantiomer content in generic formulations. (e.g., Omeprazole, a proton pump inhibitor, the S-isomer is the active form with greater bioavailability, while the R-isomer is less effective).
- Patient-side monitoring using chiral biosensors can help avoid accumulation of the undesirable isomer. (e.g., Ketamine, the S-isomer offers analgesic and antidepressant effects, whereas the R-isomer is associated with hallucinogenic side effects).
- Support sensor-assisted therapeutic drug monitoring (TDM) to improve personalised dosing (eg., Warfarin, an anticoagulant, the S-isomer is five times more potent than the R-isomer, which exhibits slower metabolism and variable therapeutic effects).
- Facilitate efficient formulation testing during batch release. (e.g., Ibuprofen, only the S-isomer is active in cyclooxygenase inhibition, while the R-isomer is inactive but can convert to the S-form).
- Aid in quality control to reduce side effects (e.g., Salbutamol, used in asthma inhalers, has a therapeutically beneficial S-isomer responsible for bronchodilation, while the R-isomer may cause adverse central nervous system effects).
5. Current challenges and future directions
Despite their promise, the transition of chiral biosensors from laboratory prototypes to commercial pharmaceutical tools is still evolving. Key challenges include:
- Achieving high enantioselectivity in complex biological samples
- Ensuring stability and reproducibility of biosensor components
- Meeting regulatory standards for analytical validation
As advancements in chiral biosensing continue to progress, exciting innovations can be anticipated.
- Integration with Lab-on-a-Chip Technologies: Advances in microfluidics and lab-on-a-chip systems will enable chiral biosensors to become even more portable and cost-effective, bringing stereoselective detection to point-of-care diagnostics.
- Miniaturization: The continued miniaturization of sensors will lead to more compact and portable devices, ideal for use in clinical settings and personal healthcare.
- Machine learning (ML) and artificial intelligence (AI) integration: The integration of AI and ML for data analysis will enhance the accuracy and speed of chiral biosensors, allowing for even more precise stereoselective detection.
6. Conclusion
Chiral biosensors are leading in a new era in pharmaceutical analysis - one where precision meets practicality. By merging selective molecular recognition with the speed and efficiency of biosensing, they offer a scalable, sustainable, and patient-focused solution to the complex challenge of enantiomer detection. As innovation continues, their adoption across the pharmaceutical value chain - from early research to manufacturing and even patient monitoring -isn’t just possible; it’s becoming essential. These smart sensors are not just tools - they’re shaping the future of safer, more effective medicines.
Reference
- Beyrami H, Golshan M, Kucińska-Lipka J, Saeb MR, Salami-Kalajahi M. Chiral-based optical and electrochemical biosensors: Synthesis, classification, mechanisms, nanostructures, and applications. Chemical Engineering Journal. 2024 Sep 10:155562.
- Ouyang YC, Yeom BJ, Zhao Y, Ma W. Progress and prospects of chiral nanomaterials for biosensing platforms. Rare Metals. 2024 Jun;43(6):2469-97.
- Xu J, Wang M, Li M, Yang J, Yang L. based chiral biosensors using enzyme encapsulation in hydrogel network for point-of-care detection of lactate enantiomers. Analytica Chimica Acta. 2023 Oct 23;1279:341834.
- Yu Z, Pan L, Ma X, Li T, Wang F, Yang D, Li M, Wang P. Detection of SARS-CoV-2 RNA with a plasmonic chiral biosensor. Biosensors and Bioelectronics. 2023 Oct 1;237:115526.
- Zhang Q, Fu M, Lu H, Fan X, Wang H, Zhang Y, Wang H. Novel potential and current type chiral amino acids biosensor based on L/D-handed double helix carbon nanotubes, polypyrrole Au nanoparticles L/D-cysteine. Sensors and Actuators B: Chemical. 2019 Oct 1;296:126667.
- Valliappan Kannappan, Chiral Drugs: A twisted tale in pharmaceuticals, 01/18/2025. .
- Valliappan Kannappan, Chiral Switch: Unlocking the potential of single enantiomers, 01/19/2025. .
- Valliappan Kannappan and Chandramouli R, Chirality in Materials Science: Designing with Handedness, 07/05/2024. .














